
Researchers Make Major Strides Towards a Vaccine Against the Zika Virus
Although absent from recent news cycles, Zika virus (ZIKV) continues to be a public health emergency and international concern. A collaborative effort between Koen Van Rompay, core scientist with the California National Primate Research Center (CNPRC), Barney Graham (Vaccine Research Institute) and Ted Pierson (National Institute of Allergy and Infectious Diseases (NIAID) brings us one step closer towards preventing another outbreak.
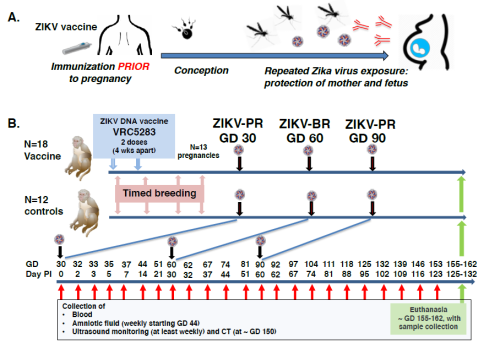
With the help of many colleagues and collaborators, the current study shows how well a ZIKV DNA vaccine called VRC5283 protects rhesus macaque offspring from viral infection. Although often unnoticed in adults, the virus can be particularly dangerous for fetuses. Infected mothers can be completely absent of symptoms but transmitting the virus to their fetus exposes them to potential health issues including: fetal death, microcephaly (abnormally small brain), and other abnormal developments. A combination of these symptoms is referred to as Congenital Zika Syndrome (CZS) and is most likely to develop when women become infected with Zika virus between the first and second trimester of pregnancy. Given the likelihood that women may not be aware of a pregnancy until many weeks post conception, researchers are working hard to develop a vaccine to protect all adolescent girls and women of child-bearing age.
Results of the current study showed lower rates of fetal loss in vaccinated animals compared to control animals exposed to the virus without vaccination.
Two of the twelve control dams lost their fetus within 30 days of conception, while none of the vaccinated dams experienced early fetal loss. Upon examination, both fetuses had clear evidence of ZIKV infection, as did nine of the ten fetuses that survived full term, indicating early transmission of the virus. Tests also showed that unvaccinated dams had detectable levels of ZIKV RNA, indicating infection, in their blood much further along in their pregnancy than vaccinated animals.
Perhaps even more exciting, none of the full-term fetuses from vaccinated dams exhibited evidence of ZIKV infection, and their brains looked more normal. Fetuses from unvaccinated dams however, displayed a range of pathology including brain lesions. Van Rompay is confident their study provides evidence that a vaccine given before conception can improve fetal outcome and he is hopeful their results can provide some of the proof needed to get the vaccine through clinical trials and out to the people who need it.
Links for open source access to original article:
Funding for this study was provided by these grants from the National Institutes of Health: R21-AI-129479-S; P51-OD-11107; R21-NS-104692; R21-HD-090856; R01-HD-098389; T32- CA009111 and by the NIAID Intramural Research Program.
Co-Authors of the study include: at UC Davis, Rebekah Keesler, Amir Ardeshir, Jennifer Watanabe, Jodie Usachenko, Anil Singapuri, Christina Cruzen, Eliza Bliss-Moreau, Ashley Murphy, JoAnn Yee, Danilo Lemos, Jackson Stuart, Erin Ball, Kevin Woolard and Lark Coffey; Helen Webster, Maria Dennis, Tulika Singh, Holly Heimsath and Sallie Permar at Duke University Medical Center; Kaitlyn Morabito, Bryant Foreman, Katherine Burgomaster, Amy Noe, Kimberly Dowd, Kathryn Foulds, Theodore Pierson, and Barney Graham at NIAID; Pietro Presicce and Suhas Kallapur, UCLA.
Written by Logan Savidge Lesavidge@ucdavis.edu
